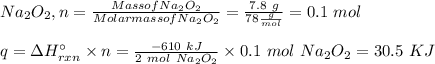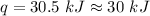Two trials are run, using excess water. In the first trial, 7.8 g of Na2O2(s) (molar mass 78 g/mol) is mixed with 3.2 g of S(s). In the second trial, 7.8 g of Na2O2(s) is mixed with 6.4 g of S(s). The Na2O2(s) and S(s) react as completely as possible. Both trials yield the same amount of SO2(aq). Which of the following identifies the limiting reactant and the heat released, q, for the two trials at 298 K?
Limiting Reactant q
A. S 30. kJ
B. S 61 kJ
C. Na2O2 30. kJ
D. Na2S2 61 kJ

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:10
There are 6.022 x 10^23 atoms of hg in 1 mole of hg. the number of atoms in 4.5 moles of hg can be found by multiplying 4.5 by 6.022 x 10^23 a. 2.7 x 10^24 b. 27 x 10^23 c. 2.71 x10^24 d. 27.099 x 10^23
Answers: 3

Chemistry, 22.06.2019 00:30
The clouds are grey and ground is wet. a quantitative b qualitative
Answers: 1

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 1

Chemistry, 22.06.2019 09:40
Consider this initial-rate data at a certain temperature for the reaction described by
Answers: 1
You know the right answer?
Two trials are run, using excess water. In the first trial, 7.8 g of Na2O2(s) (molar mass 78 g/mol)...
Questions

Geography, 05.04.2021 16:50



Engineering, 05.04.2021 16:50


Mathematics, 05.04.2021 16:50

Business, 05.04.2021 16:50


Biology, 05.04.2021 16:50

History, 05.04.2021 16:50




Biology, 05.04.2021 16:50


Biology, 05.04.2021 16:50

Mathematics, 05.04.2021 16:50



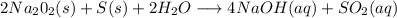
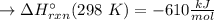
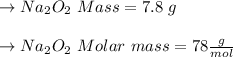
 Has been the reactant which is limited since the two experiments are equal to
Has been the reactant which is limited since the two experiments are equal to