
Chemistry, 20.03.2021 19:20 grosst4333
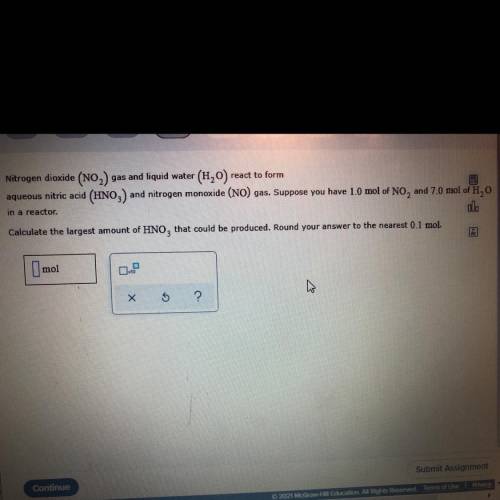
Nitrogen dioxide (NO2) gas and liquid water (H20) react to form
aqueous nitric acid (HNO3) and nitrogen monoxide (NO) gas. Suppose you have 1.0 mol of NO, and 7.0 mol of H, 0
olle
Calculate the largest amount of HNO, that could be produced. Round your answer to the nearest 0.1 mol.
in a reactor.
Imo1
x
?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Of the groups of elements below, which are most likely to gain electrons to become anions? a. alkali metal b. boron group c. halogen d. transition metal
Answers: 2

Chemistry, 23.06.2019 01:30
Which statement accurately represents the arrangement of electrons in bohr’s atomic model?
Answers: 2

Chemistry, 23.06.2019 04:20
The reaction below shows a system in equilibrium. how would a decrease in temperature affect this reaction? a. the rate of formation of the gases would increase. b. the equilibrium of the reaction would shift to the left. c. the equilibrium would shift to cause the gases to sublime into solids. d. the chemicals on the left would quickly form the chemical on the right.
Answers: 1

Chemistry, 23.06.2019 13:00
Which of the following statements is true about both nuclear fusion and nuclear fission? they occur in the sun. heavy atoms are split. two light nuclei combine. some mass changes into energy.
Answers: 1
You know the right answer?
Nitrogen dioxide (NO2) gas and liquid water (H20) react to form
aqueous nitric acid (HNO3) and nitr...
Questions

English, 05.02.2020 05:44


Chemistry, 05.02.2020 05:44





Mathematics, 05.02.2020 05:44

Mathematics, 05.02.2020 05:44


Biology, 05.02.2020 05:44


Chemistry, 05.02.2020 05:44

English, 05.02.2020 05:44






Mathematics, 05.02.2020 05:45



