Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
What is the molecular formula for a compound that is 46.16% carbon, 5.16% hydrogen, and 48.68% fluorine? the molar mass of the compound is 156.12 g/mol
Answers: 2



Chemistry, 22.06.2019 19:10
Which statement correctly describes the phosphate ion, ? it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge on the phosphorus atom. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge on the phosphorus atom.
Answers: 3
You know the right answer?
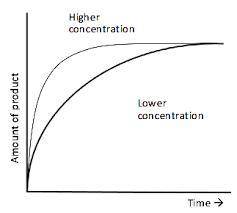
Describe what is happening at each part of the graph, include; number of reactant particles, frequen...
Questions







Mathematics, 22.04.2020 04:43

Mathematics, 22.04.2020 04:43




Chemistry, 22.04.2020 04:43

Business, 22.04.2020 04:44


Mathematics, 22.04.2020 04:44


Computers and Technology, 22.04.2020 04:44



Mathematics, 22.04.2020 04:44