
Chemistry, 29.09.2019 15:30 Fireburntbudder
Which of these elements can become stable by losing 1 electron? explain
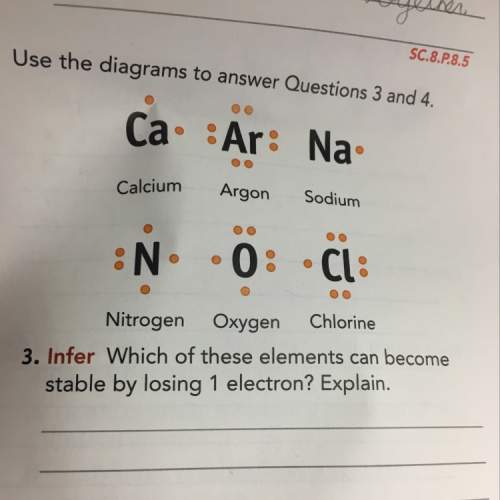

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

You know the right answer?
Which of these elements can become stable by losing 1 electron? explain
...
...
Questions


Business, 20.10.2020 14:01

Business, 20.10.2020 14:01


Advanced Placement (AP), 20.10.2020 14:01


Mathematics, 20.10.2020 14:01

Biology, 20.10.2020 14:01



History, 20.10.2020 14:01

Social Studies, 20.10.2020 14:01



English, 20.10.2020 14:01

Biology, 20.10.2020 14:01

Mathematics, 20.10.2020 14:01

Health, 20.10.2020 14:01

Mathematics, 20.10.2020 14:01

English, 20.10.2020 14:01



