
Chemistry, 22.03.2021 21:40 hardwick744
The solubility product for Zn(OH)2 is 3.0×10−16. The formation constant for the hydroxo complex, Zn(OH)2−4, is 4.6×1017. What is the minimum concentration of OH− required to dissolve 1.7×10−2 mol of Zn(OH)2 in a liter of solution?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:30
(1.6 × 10-19)(5.0 × 106) = c × 10d identify the missing numbers below to show the result of multiplying the numbers.
Answers: 1

Chemistry, 22.06.2019 08:00
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 11:00
The diagram below shows the different phase transitions that occur in matter. which arrow represents the transition in which dew is formed?
Answers: 1

Chemistry, 22.06.2019 13:00
6. using 3 – 4 sentences explain (in your own words) why water expands when it freezes? 7. using your knowledge of colligative properties explain whether sodium chloride or calcium chloride would be a more effective substance to melt the ice on a slick sidewalk. use 3 – 4 sentences in your explanation.
Answers: 1
You know the right answer?
The solubility product for Zn(OH)2 is 3.0×10−16. The formation constant for the hydroxo complex, Zn(...
Questions

Mathematics, 18.04.2020 19:26

Mathematics, 18.04.2020 19:26


English, 18.04.2020 19:26


Mathematics, 18.04.2020 19:26

Computers and Technology, 18.04.2020 19:26



Health, 18.04.2020 19:26


Mathematics, 18.04.2020 19:26

Mathematics, 18.04.2020 19:27

Mathematics, 18.04.2020 19:27


Arts, 18.04.2020 19:27


History, 18.04.2020 19:27

Mathematics, 18.04.2020 19:27

![[OH^-]= 1.12 \times 10^{-2} \ M](/tpl/images/1212/0889/be159.png)


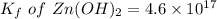
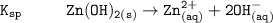
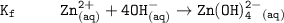
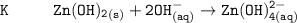
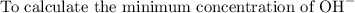
![K = \dfrac{Zn(OH)^-_4}{[OH^-]^2} \\ \\](/tpl/images/1212/0889/c5800.png)
![[OH^-]^2= \dfrac {0.017}{1.380 \times 10^2}](/tpl/images/1212/0889/33df3.png)
![[OH^-]^2=1.232 \times 10^{-4}](/tpl/images/1212/0889/f97e0.png)


