
Chemistry, 22.03.2021 22:00 carterlewis02
1) In the picture , which of the following is the best evidence that a chemical reaction has occurred?
The mass is 2402.0 g
The phenol red is a yellow color
Bubbles are formed in the inverted tube
There is no evidence of a reaction
2) The reaction depicted in question 1 is: 2 H2O2(aq) → 2 H2O(l) + 1 O2(g)
A) Give the formula of a product.
B) What is the phase of matter for O2?
C) If the reaction were performed in a closed container and the mass was measured to be 2402.0 g, what would you expect the mass to be after the reaction is complete? Why?
3. It is possible to use dimensional analysis to calculate the number of molecules in 3.2 moles of carbon dioxide if we use the fact that 1 mole = 6.02 x 10^23 molecules. Which of the following equations is the correct way to set up this calculation?
(see attachment 2)
4) Write 602000000000 in scientific notation.
5) Balance and name the type of reaction (synthesis, decomposition, single displacement, double displacement or combustion) for each of the following:
A) ___ I2 + ___ F2 → ___ IF7
Type of rxn:
B) ___ Sn + ___ Cr2O3 → ___ Cr + ___ SnO2
Type of rxn:
C) ___ C9H20 + ___ O2 → ___ CO2 + ___ H2O
Type of rxn:
6) Determine the molar mass for each of the following
A) Cr2O3 B) IF7
7) Determine the mass of 0.804 moles Cr2O3.
8) Calculate the number of moles in 76.5 grams of IF7.

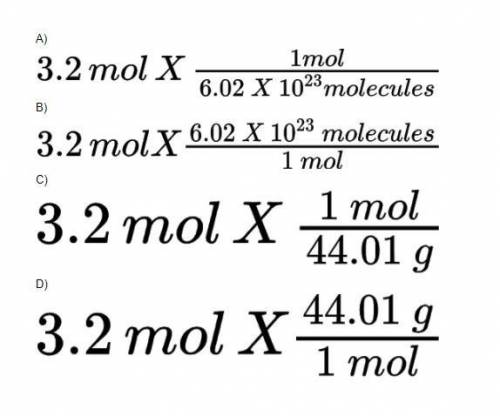

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 12:30
Select the correct answer. what is the nature of the se-cl bond in a molecule of selenium chloride (secl2) if the electronegativity value of selenium is 2.55 and that of chlorine is 3.16?
Answers: 3


Chemistry, 21.06.2019 16:30
Energy is released during which phase changes? check all that apply. boiling condensing depositing freezing melting subliming
Answers: 2

Chemistry, 21.06.2019 23:00
At room temperature what happens to the average kinetic energy of the molecules of a solid, liquid, and a gas
Answers: 2
You know the right answer?
1) In the picture , which of the following is the best evidence that a chemical reaction has occurre...
Questions

Mathematics, 14.10.2019 21:00




Social Studies, 14.10.2019 21:00

English, 14.10.2019 21:00


History, 14.10.2019 21:00

English, 14.10.2019 21:00

Mathematics, 14.10.2019 21:00






Chemistry, 14.10.2019 21:00

Computers and Technology, 14.10.2019 21:00


History, 14.10.2019 21:00



