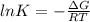The value of the equilibrium constant for a reaction
a. changes with temperature.
b. changes...

Chemistry, 23.03.2021 07:30 orlando19882000
The value of the equilibrium constant for a reaction
a. changes with temperature.
b. changes with time.
c. is the same under all conditions.
d. changes with concentration.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:40
What kind of ion is contained in salts that produce an acidic solution? a positive ion that attracts a proton from water a positive ion that releases a proton to water a negative ion that attracts a proton from water a negative ion that releases a proton to water
Answers: 1

Chemistry, 21.06.2019 21:00
Iwll give extra points to who gets this for ! what type of reaction is this? ?
Answers: 2

Chemistry, 22.06.2019 02:40
Consider the nuclear equation below. 239/94 pu—-> x+ 4/2 he. what is x?
Answers: 2

Chemistry, 22.06.2019 03:30
If a solution is considered basic, then a) the hydroxide ion and hydronium ion concentrations are equal. b) the hydroxide ion concentration is less than the hydronium ion concentration. c) the hydronium ion concentration is greater than the hydroxide ion concentration. d) the hydroxide ion concentration is greater than the hydronium ion concentration.
Answers: 1
You know the right answer?
Questions




Mathematics, 29.10.2020 01:00



Mathematics, 29.10.2020 01:00


Mathematics, 29.10.2020 01:00




Advanced Placement (AP), 29.10.2020 01:00



Mathematics, 29.10.2020 01:00

Mathematics, 29.10.2020 01:00

Mathematics, 29.10.2020 01:00

Mathematics, 29.10.2020 01:00