What is the freezing point in °C of a 2.20 molal solution of lithium bromide in water?
...

Chemistry, 23.03.2021 17:30 ashlpiriz123
What is the freezing point in °C of a 2.20 molal solution of lithium bromide in water?
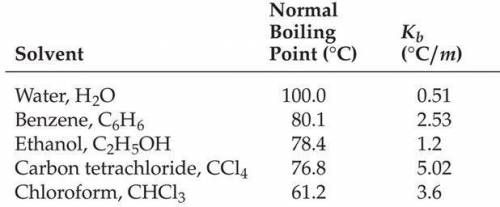

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:50
How many liters of oxygen gas, at standard temperature and pressure, will react with 35.8 grams of iron metal? 4 fe (s) + 3 o₂ (g) → 2 fe₂o₃ (s)
Answers: 2

Chemistry, 22.06.2019 13:30
Table sugar completely dissolved in water is an example of a?
Answers: 1


Chemistry, 22.06.2019 23:30
The density of the solid phase of a substance is 0.90 g/cm3 and the density of the liquid phase is 1.0 g/cm3. a large increase in pressure will a. lower the freezing point b. raise the freezing point c. lower the boiling point d. raise the triple point e. lower the triple point
Answers: 1
You know the right answer?
Questions






Mathematics, 15.04.2020 02:48




Mathematics, 15.04.2020 02:48








Computers and Technology, 15.04.2020 02:49




