Instructions
Use the following chemical reaction and masses to answer the following questions
...

Instructions
Use the following chemical reaction and masses to answer the following questions
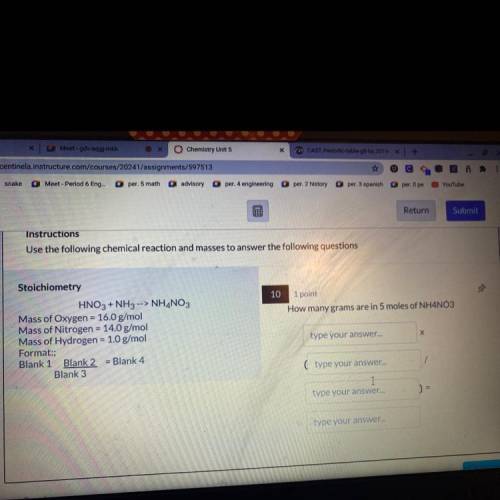
10 1 point
How many grams are in 5 moles of NH4NO3
Stoichiometry
HNO3 + NH3 --> NH4NO3
Mass of Oxygen = 16.0 g/mol
Mass of Nitrogen = 14.0 g/mol
Mass of Hydrogen = 1.0 g/mol
Format:;
Blank 1 Blank 2 = Blank 4
Blank 3
type your answer...
Х
(type your answer...
I
type your answer...
type your answer...
PLEASE HELP

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 18:00
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3

Chemistry, 23.06.2019 00:30
There are approximately 15 milliliters (ml) in 1 tablespoon (tbsp). what expression can be used to find the approximate number of milliliters in 3 tbsp?
Answers: 1

Chemistry, 23.06.2019 02:40
Calculate the standard enthalpy of formation of liquid methanol, ch3oh(l), using the following information: c(graphite) + o2 latex: \longrightarrow ⟶ co2(g) latex: \delta δ h° = –393.5 kj/mol h2(g) + o2 latex: \longrightarrow ⟶ h2o(l) latex: \delta δ h° = –285.8 kj/mol ch3oh(l) + o2(g) latex: \longrightarrow ⟶ co2(g) + 2h2o(l) latex: \delta δ h° = –726.4 kj/mol
Answers: 3
You know the right answer?
Questions



History, 30.01.2020 02:44


English, 30.01.2020 02:44


Mathematics, 30.01.2020 02:44

Social Studies, 30.01.2020 02:44

History, 30.01.2020 02:44



English, 30.01.2020 02:44







English, 30.01.2020 02:44




