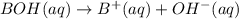Chemistry, 25.03.2021 23:40 cuppykittyy
According to the Arrhenius concept, an acid is a substance that . According to the Arrhenius concept, an acid is a substance that . tastes bitter can accept a pair of electrons to form a coordinate covalent bond is capable of donating one or more H reacts with the solvent to form the cation formed by autoionization of that solvent causes an increase in the concentration of H in aqueous solutions

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:30
If i make a solution by adding 83grams of sodium hydroxide to 750ml i’d water what is the molarity of sodium hydroxide
Answers: 1

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

Chemistry, 22.06.2019 08:40
Ageologist determines that a sample of a mineral can't be scratched by a steel nail but can be scratched by a masonry drill bit. based on this information, the sample mineral has to be softer than a. orthoclase. b. fluorite. c. apatite. d. corundum.
Answers: 2

You know the right answer?
According to the Arrhenius concept, an acid is a substance that . According to the Arrhenius concept...
Questions

Mathematics, 03.03.2021 14:00


History, 03.03.2021 14:00


History, 03.03.2021 14:00

Mathematics, 03.03.2021 14:00



English, 03.03.2021 14:00

Biology, 03.03.2021 14:00


Mathematics, 03.03.2021 14:00

Mathematics, 03.03.2021 14:00

Mathematics, 03.03.2021 14:00

English, 03.03.2021 14:00



Biology, 03.03.2021 14:00

Mathematics, 03.03.2021 14:00

Chemistry, 03.03.2021 14:00