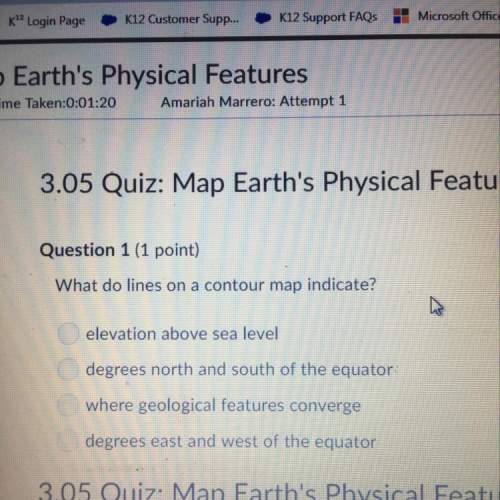
Chemistry, 26.03.2021 14:00 chrissycherry
C3H8+5O2 →3CO2+4H20, what is the maximum mass of CO2 which could be formed from 5.52 g of C3H8 and 8.03 g of O2?Answer in units of g.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:30
At a temperature of 393 k, the temperature of a sample of nitrogen is 1.07 atm what will the pressure be at a temperature of 478 k
Answers: 1

Chemistry, 22.06.2019 07:30
Label a-f based on the table using c for concentrated and d for dilute
Answers: 2

Chemistry, 23.06.2019 10:00
Lord kelvin described the concept of absolute zero temperature and the laws relating to the change of thermal energy during chemical reactions what type of chemist would he be considered today
Answers: 1

Chemistry, 23.06.2019 19:30
How many kmin (kilominutes) are there in 18 weeks and 5 days?
Answers: 3
You know the right answer?
C3H8+5O2 →3CO2+4H20,
what is the maximum mass of CO2 which could be formed from 5.52 g of C3H8 and...
Questions


English, 18.10.2019 00:10







Mathematics, 18.10.2019 00:10

History, 18.10.2019 00:10

History, 18.10.2019 00:10









Computers and Technology, 18.10.2019 00:20