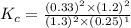Chemistry, 26.03.2021 21:30 ariellake8551
Nitrogen and water react to form nitrogen monoxide and hydrogen, like this: N2(g) + 2H2O(g) → 2NO(g) +2H2(g)Also, a chemist finds that at a certain temperature the equilibrium mixture of nitrogen, water, nitrogen monoxide, and hydrogen has the following composition: compound pressure at equilibrium N2 0.25 M H20 1.3 M NO 0.33 M H2 1.2 MCalculate the value of the equilibrium constant for this reaction. Round your answer to significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 16:30
How many atoms of oxygen are contained in 160 grams of n2o3
Answers: 2

Chemistry, 22.06.2019 06:00
How much would the freezing point of water decrease if 4 mol of sugar were added to 1 kg of water(k=1.86 c/mol/kg for water and i=1 for sugar
Answers: 1

Chemistry, 22.06.2019 18:10
Areader can tell that the meaning of “obnoxious” will include “having the quality of something” because of the .a) prefix b)pronunciation c)suffix d) word root
Answers: 3

Chemistry, 22.06.2019 22:30
Is the idea of spontaneous generation supported by redi's experiment? justify your answer in 2-3 sentences?
Answers: 1
You know the right answer?
Nitrogen and water react to form nitrogen monoxide and hydrogen, like this: N2(g) + 2H2O(g) → 2NO(g)...
Questions


Mathematics, 28.04.2021 17:00

Biology, 28.04.2021 17:00



Spanish, 28.04.2021 17:00

Mathematics, 28.04.2021 17:00


Mathematics, 28.04.2021 17:00


Mathematics, 28.04.2021 17:00






Mathematics, 28.04.2021 17:00


Mathematics, 28.04.2021 17:00

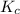
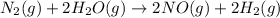
![K_c=\frac{[NO]^2\times [H_2]^2}{[H_2O]^2\times [N_2]^1}](/tpl/images/1224/3364/cca59.png)