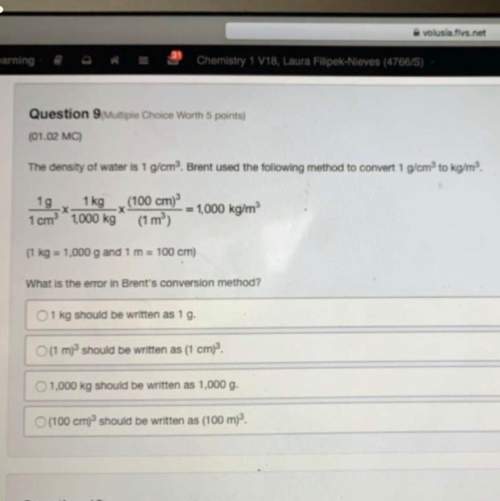
An ideal gas fills a balloon at a temperature of 227ºC and standard pressure. By what factor will the volume of the balloon change if the gas in the balloon is heated to 327ºC at standard pressure? Identify which gas law applies and show your work to receive full credit. Highlight the correct answer below. (2 points)
a. 227/327
b. 5/6
c. 6/5
d. 2/1
e. 327/227

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 02:30
Margaret wants to make an orange flavored drink by stirring powdered drink mix into a glass of water. she doesn't like drinks that have small clumps of powdered solid in them, so she wants the drink to be a perfect solution. what factors should margaret not consider when deciding how much powder to add to her glass of water?
Answers: 3

Chemistry, 22.06.2019 07:30
What is i fracture in the crust called when land move up, down or sideways
Answers: 2

Chemistry, 22.06.2019 09:00
At 300 mm hg, a gas has a volume of 380 l, what is the volume at standard pressure
Answers: 1
You know the right answer?
An ideal gas fills a balloon at a temperature of 227ºC and standard pressure. By what factor will th...
Questions

Mathematics, 23.08.2019 16:50



Biology, 23.08.2019 16:50




History, 23.08.2019 16:50

Social Studies, 23.08.2019 16:50

History, 23.08.2019 16:50





Computers and Technology, 23.08.2019 16:50

Chemistry, 23.08.2019 16:50


Mathematics, 23.08.2019 16:50