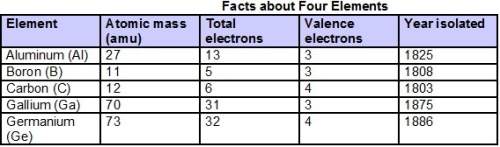
Chemistry, 28.03.2021 21:10 money2tymes
. How is stoichiometry used to calculate amounts of substances in a chemical reaction? (1 point) b. Use the balanced equation to answer the following questions. (2 points) CuSO4(aq) + 2NaOH(aq) Cu(OH)2(s) + Na2SO4(aq)
i. What is the ratio of moles of CuSO4 to moles of NaOH? (.5 point)
ii. If 638.44 g CuSO4 reacts with 240.0 NaOH, which is the limiting reagent? (.5 point)
iii. Use the limiting reagent to determine how many grams of Cu(OH)2 should precipitate out in the reaction. (.5 point)
iv. If only 174.6 g of Cu(OH)2 precipitate were actually collected from the reaction, what would the percent yield be? (.5 point)

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Which type of reaction always has an element and a compound as reactants
Answers: 1

Chemistry, 22.06.2019 05:30
Why is soap used to remove grease? a. its nonpolar end dissolves the grease. b. it makes the water bond with the grease. c. it chemically bonds with the grease. d. its polar end dissolves the grease.correct answer for apex - a, its nonpolar end dissolves the grease.
Answers: 1

Chemistry, 22.06.2019 09:00
The nuclear fission process releases neutrons and question 27 options: alpha particles electrons energy beta particles
Answers: 1

Chemistry, 22.06.2019 18:10
Measurements that have similar values are: a. usually accurate b. sometimes accurate c. always accurate d. never accurate
Answers: 1
You know the right answer?
. How is stoichiometry used to calculate amounts of substances in a chemical reaction? (1 point) b....
Questions

Social Studies, 28.01.2021 23:00




SAT, 28.01.2021 23:00

Mathematics, 28.01.2021 23:00



Geography, 28.01.2021 23:00

Spanish, 28.01.2021 23:00

Mathematics, 28.01.2021 23:00

Mathematics, 28.01.2021 23:00

Health, 28.01.2021 23:00

Mathematics, 28.01.2021 23:00

Computers and Technology, 28.01.2021 23:00

English, 28.01.2021 23:00

Computers and Technology, 28.01.2021 23:00

Mathematics, 28.01.2021 23:00

History, 28.01.2021 23:00