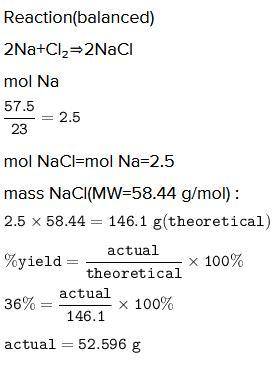Chemistry, 29.03.2021 19:20 joyceslater16
A chemist reacted 57.50 grams of sodium metal with an excess amount of chlorine gas. The chemical reaction that occurred is shown.
Na + Cl2 → NaCl
If the percentage yield of the reaction is 86%, what is the actual yield? Show your work, including the use of stoichiometric calculations and conversion factors.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3


Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3

Chemistry, 22.06.2019 21:20
40dm3 of gas at 760 torr are heated from 5°c to 50°c what is the new volume
Answers: 3
You know the right answer?
A chemist reacted 57.50 grams of sodium metal with an excess amount of chlorine gas. The chemical re...
Questions



Mathematics, 12.11.2020 18:10


Mathematics, 12.11.2020 18:10




Mathematics, 12.11.2020 18:10

Biology, 12.11.2020 18:10

Arts, 12.11.2020 18:10

Mathematics, 12.11.2020 18:10


Biology, 12.11.2020 18:10

English, 12.11.2020 18:10

Chemistry, 12.11.2020 18:10

Mathematics, 12.11.2020 18:10

French, 12.11.2020 18:10


Mathematics, 12.11.2020 18:10