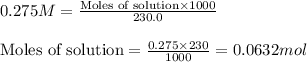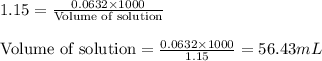Chemistry, 24.09.2019 01:50 krystalhurst97
A230.0-ml sample of a 0.275 m solution is left on a hot plate overnight; the following morning the solution is 1.15 m. what volume of solvent has evaporated from the 0.275 m solution?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:50
If the mass of the products measured 120g what would the mass of the reactants a. 30g b. 60g c. 120g d. 240g
Answers: 1

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 10:30
How do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 2

Chemistry, 22.06.2019 13:00
What is the mass of 2.00 l of an intravenous glucose solution with a density of 1.15 g/ml?
Answers: 2
You know the right answer?
A230.0-ml sample of a 0.275 m solution is left on a hot plate overnight; the following morning the...
Questions



















Advanced Placement (AP), 24.10.2019 23:43

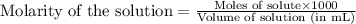 .....(1)
.....(1)