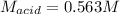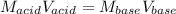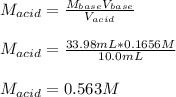Suppose you are titrating vinegar, which is an acetic acid solution of unknown strength, with a sodium hydroxide solution according to the equation H C 2 H 3 O 2 + N a O H ⟶ H 2 O + N a C 2 H 3 O 2 HCX2HX3OX2+NaOH⟶HX2O+NaCX2HX3OX2 If you require 33.98 mL of 0.1656 M N a O H NaOH solution to titrate 10.0 mL of H C 2 H 3 O 2 HCX2HX3OX2 solution, what is the molar concentration of acetic acid in the vinegar? Type

Answers: 2
Another question on Chemistry



Chemistry, 22.06.2019 22:00
4.25g sample of solid ammonium nitrate dissolves in 60.0g of water in a coffee-cup calorimeter, the temperature drops from 22.0 c to 16.9 c. assume that the specific heat of the solution is the same as that of pure water. calculate delta(h) (in kj/mol nh4no3) for the solution proces.
Answers: 2

Chemistry, 23.06.2019 18:30
Select the correct answer. which statement best explains the octet rule? a. atoms gain, lose, or share electrons to achieve a full valence shell. b. compounds always consist of eight electrons. c. molecules always consist of eight atoms. d. all atoms require eight electrons to form bonds.
Answers: 2
You know the right answer?
Suppose you are titrating vinegar, which is an acetic acid solution of unknown strength, with a sodi...
Questions

English, 03.02.2020 04:59

Biology, 03.02.2020 04:59


Geography, 03.02.2020 04:59

English, 03.02.2020 04:59




Mathematics, 03.02.2020 04:59

History, 03.02.2020 04:59


History, 03.02.2020 04:59


English, 03.02.2020 04:59

Biology, 03.02.2020 04:59



History, 03.02.2020 04:59