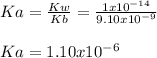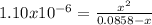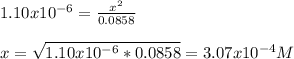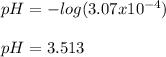Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:00
What is the molality of a solution that has 4 mol of kci in 0.800 kg of water
Answers: 3

Chemistry, 21.06.2019 17:30
What are the major products produced in the combustion of c10h22 under the following conditions? write balanced chemical equations for each. a. an excess of oxygen b. a slightly limited oxygen supply c. a very limited supply of oxygen d. the compound is burned in air
Answers: 2


Chemistry, 22.06.2019 13:00
One of the hopes for solving the world's energy problem is to make use of the fusion reaction 21h +31h --> 42he + 10n + energy how much energy is released when 1 mol of deuterium is fused with 1 mol of tritium according to the above reaction? the masses of the atoms and the neutrons are as follows: 21h = 2.0140 amu 31h = 3.01605 amu 42he = 4.002603 amu 10n = 1.008665 amu. the speed of light is 2.9979 x 108 m/s.
Answers: 1
You know the right answer?
Calculate the pH at the equivalence point when 22.0 mL of 0.200 M hydroxylamine, HONH2, is titrated...
Questions

English, 19.11.2019 22:31

Mathematics, 19.11.2019 22:31

Chemistry, 19.11.2019 22:31

Mathematics, 19.11.2019 22:31





Biology, 19.11.2019 22:31






Mathematics, 19.11.2019 22:31

English, 19.11.2019 22:31

Mathematics, 19.11.2019 22:31



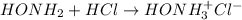
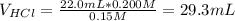
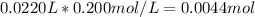
![[HONH_3^+Cl^-]=\frac{0.0044mol}{0.022L+0.0293L} =0.0858M](/tpl/images/1235/6200/36779.png)

![Ka=\frac{[H_3O^+][HONH_2]}{[HONH_3^+]}](/tpl/images/1235/6200/cfc73.png)