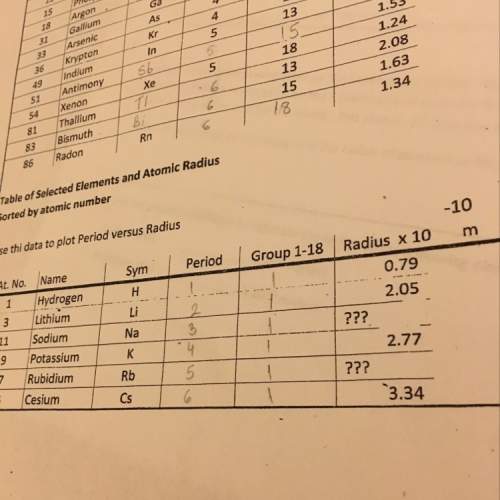
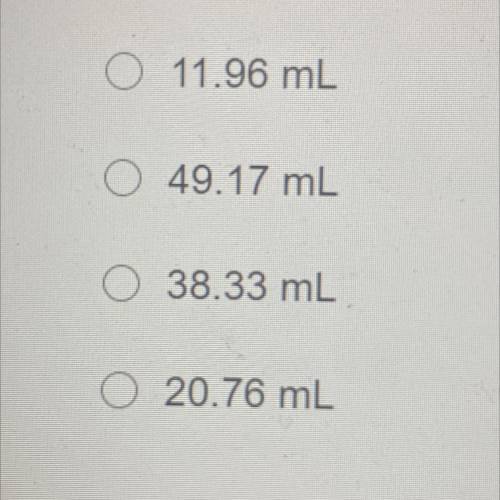
An unopened soda bottle contains 46.0mL of gas confined at a pressure of 1.30 atm at a temperature of 5.0°C. If the bottle is
dropped into a lake and sinks to a depth at which the pressure is 2.85 atm and temperature is 2.09°C, what will the volume of the
gas in the bottle be?

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 17:30
Oil rich countries in the middle east cover about 4% of earths total land area but prossess about 48% of the worlds known oil reserves what is the main reason for high concentration of reserves in this part of the world
Answers: 3

Chemistry, 23.06.2019 03:00
Asample of sea water contains 6.28g of sodium chloride per litre of solution. how many milligrams of sodium chloride would be contained in 15.0ml of this solution?
Answers: 3

Chemistry, 23.06.2019 05:40
Convert a speed of 201 cm/s to units of inches per minute. also, show the unit analysis by dragging components into the unit‑factor slots.
Answers: 1
You know the right answer?
An unopened soda bottle contains 46.0mL of gas confined at a pressure of 1.30 atm at a temperature o...
Questions

Computers and Technology, 21.04.2020 01:16




Mathematics, 21.04.2020 01:16


Mathematics, 21.04.2020 01:16

English, 21.04.2020 01:16

English, 21.04.2020 01:16

Mathematics, 21.04.2020 01:16



Advanced Placement (AP), 21.04.2020 01:17


Mathematics, 21.04.2020 01:17



Health, 21.04.2020 01:17