Chemistry, 02.04.2021 02:00 KieraKimball
Ammonium phosphate NH43PO4 is an important ingredient in many solid fertilizers. It can be made by reacting aqueous phosphoric acid H3PO4 with liquid ammonia. Calculate the moles of ammonium phosphate produced by the reaction of 0.050mol of phosphoric acid. Be sure your answer has a unit symbol, if necessary, and round it to the correct number of significant digits.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:30
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1

Chemistry, 22.06.2019 01:00
What are the variables in gay-lussac’s law? pressure and volume pressure, temperature, and volume pressure and temperature volume, temperature, and moles of gas
Answers: 1

Chemistry, 22.06.2019 01:30
Aroller coaster car is traveling down a track at 22 m/s. the car has a mass of 2000 kg. what is the kinetic energy of the car? a) 22,000 j b) 968,000 j c) 484,000 j d) 44,000 j
Answers: 2

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 1
You know the right answer?
Ammonium phosphate NH43PO4 is an important ingredient in many solid fertilizers. It can be made by r...
Questions


Chemistry, 13.10.2020 14:01

Mathematics, 13.10.2020 14:01

Mathematics, 13.10.2020 14:01

Mathematics, 13.10.2020 14:01

Advanced Placement (AP), 13.10.2020 14:01

Social Studies, 13.10.2020 14:01

Spanish, 13.10.2020 14:01



History, 13.10.2020 14:01




History, 13.10.2020 14:01


Mathematics, 13.10.2020 14:01

Mathematics, 13.10.2020 14:01


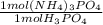 = 0.050 mol (NH₄)₃PO₄
= 0.050 mol (NH₄)₃PO₄