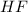Hydrogen fluoride reacts with ammonia in an acid-base reaction:
HF (aq) + NH3 (aq) -->
<...

Chemistry, 05.04.2021 07:50 SearchLight
Hydrogen fluoride reacts with ammonia in an acid-base reaction:
HF (aq) + NH3 (aq) -->
a. Determine the products made by completing the reaction above.
b. Identify and label one of the conjugate acid-base pairs.
Please I will give brainliest I’m stuck

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 12:00
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1


Chemistry, 22.06.2019 22:00
The diagrams to the right show the distribution and arrangement of gas particles in two different containers. according to kinetic-molecular theory, which of the following statements is true? check all that apply. if the temperatures of both containers are equal, container a has greater pressure than container b. if the volume of container a decreased, its pressure would decrease. if the pressure in both containers is equal, container a has a lower temperature than container b.
Answers: 2

You know the right answer?
Questions


Mathematics, 15.04.2021 16:30

English, 15.04.2021 16:30









Mathematics, 15.04.2021 16:30



English, 15.04.2021 16:30

Mathematics, 15.04.2021 16:30

Mathematics, 15.04.2021 16:30

Biology, 15.04.2021 16:30


Mathematics, 15.04.2021 16:30

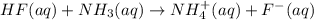
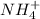 : conjugate acid of
: conjugate acid of 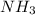
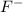 : conjugate base of
: conjugate base of