Consider the following balanced equation:
2KCIO3(s) → 2KCl(s) + 302(g)
How many moles of O2 w...

Chemistry, 06.04.2021 23:20 hncriciacheichi
Consider the following balanced equation:
2KCIO3(s) → 2KCl(s) + 302(g)
How many moles of O2 will be obtained by decomposing
3.50 moles of KCIO3?
0.530 mole
O 3.00 moles
O 2.30 moles
5.25 moles

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 11:50
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3

Chemistry, 22.06.2019 14:30
Which of the following represents the ester functional group? a. -coo- b. -cho c. -cooh d. c=o
Answers: 1

Chemistry, 22.06.2019 23:00
Movement that is like a t a type of wave that transfers energy where the particles in the medium move in a circle motion while the energy travels left or right. a type of wave that transfers energy where the particles in the medium move perpendicular to the direction in which the energy is traveling. transfers energy from one location to another a type of wave that transfers energy where the particles in the medium move parallel to the direction in which the energy is traveling. movement that is back and forth, like an equal sign = 1. wave 2. parallel movement 3. perpendicular movement 4. transverse wave 5. longitudinal wave 6. surface wave
Answers: 1

Chemistry, 22.06.2019 23:00
What is the average rate of the reaction between 10 and 20 s?
Answers: 1
You know the right answer?
Questions

History, 09.12.2020 01:00




Mathematics, 09.12.2020 01:00


History, 09.12.2020 01:00

Mathematics, 09.12.2020 01:00


Biology, 09.12.2020 01:00


Arts, 09.12.2020 01:00

Mathematics, 09.12.2020 01:00

History, 09.12.2020 01:00

Mathematics, 09.12.2020 01:00





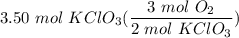 [DA} Multiply [Cancel out units]:
[DA} Multiply [Cancel out units]: 


