
Chemistry, 08.04.2021 22:20 jungcoochie101
g Consider a pure sample of a radioactive isotope with a mass number of (46 A). If the sample has mass of (25.0 B) micrograms and the isotope has a half-life of (4.50 C)x106 years, determine the decay rate for the sample. Give your answer in decays/second and with 3 significant figures.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:30
You are performing an experiment in a lab to attempt a new method of producing pure elements from compounds. the only problem is that you do not know what element will form. by your previous calculations you know that you will have 6.3 moles of product. when it is complete, you weigh it and determine you have 604.4 grams. what element have you produced?
Answers: 1

Chemistry, 22.06.2019 15:00
Answer explain why it is not possible to deduce a complete order of reactivity.
Answers: 3


Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium
Answers: 1
You know the right answer?
g Consider a pure sample of a radioactive isotope with a mass number of (46 A). If the sample has ma...
Questions

Mathematics, 16.10.2019 22:10

History, 16.10.2019 22:10

Health, 16.10.2019 22:10




History, 16.10.2019 22:10





History, 16.10.2019 22:10


English, 16.10.2019 22:10

German, 16.10.2019 22:10





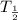 = ( 4.50 + C ) × 10⁶ years
= ( 4.50 + C ) × 10⁶ years 

