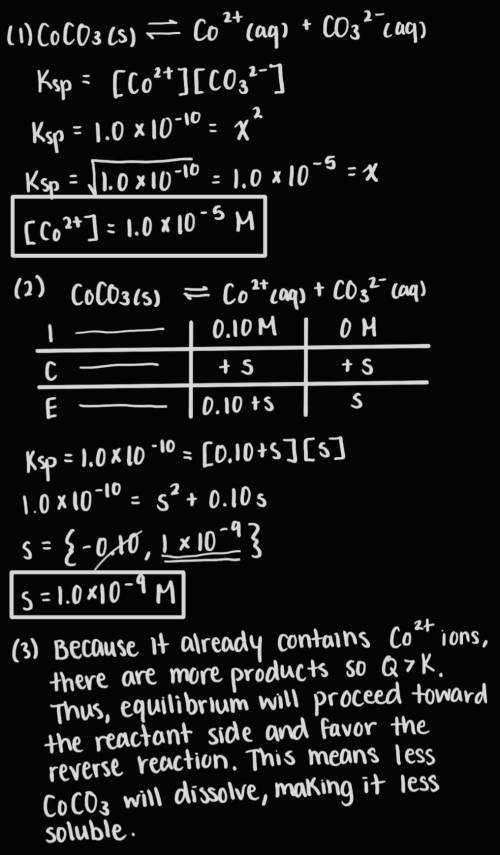Chemistry, 09.04.2021 23:50 valoiserika1229
Answer the following questions about the solubility of CoCO3(s). The value of Ksp for CoCO3(s) is 1.0 × 10^−10.
A. Calculate the value of [Co2+] in a saturated solution of CoCO3 in distilled water.
B. If 0.10 M of Co2+ is already present in distilled water, calculate the molar solubility of CoCO3(s).
C. Explain why CoCO3 is less soluble in distilled water that already contains Co2+

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:10
Which of the following elements would you expect to have the lowest ionization energy value? fluorine, lithium, neon, nitrogen
Answers: 2

Chemistry, 22.06.2019 06:00
How many atoms of mg are present in 97.22 grams of mg? 6.022 × 1023 2.408 × 1024 4.818 × 1024 5.855 × 1025
Answers: 2

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3
You know the right answer?
Answer the following questions about the solubility of CoCO3(s). The value of Ksp for CoCO3(s) is 1....
Questions



Chemistry, 01.10.2019 13:10

English, 01.10.2019 13:10



Computers and Technology, 01.10.2019 13:10

History, 01.10.2019 13:10

Biology, 01.10.2019 13:10


Biology, 01.10.2019 13:10

English, 01.10.2019 13:10

English, 01.10.2019 13:10

Mathematics, 01.10.2019 13:10


Mathematics, 01.10.2019 13:10



History, 01.10.2019 13:10

Mathematics, 01.10.2019 13:10

![K = \frac{[products]}{[reactants]}](/tpl/images/1250/0658/0c10f.png) increases.
increases.