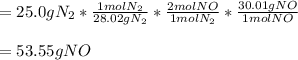Chemistry, 10.04.2021 07:20 aubreymoore9441
Using the balanced equation N2+O2=2NO, how many grams of NO can be produced when 25.0 grams of N react?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3

Chemistry, 22.06.2019 12:00
Give the set of reactants (including an alkyl halide and a nucleophile) that could be used to synthesize the following ether: draw the molecules on the canvas by choosing buttons from the tools (for bonds and charges), atoms, and templates toolbars, including charges where needed. ch3ch2och2ch2chch3 | ch3
Answers: 1

Chemistry, 22.06.2019 12:30
If 22.5 liters of oxygen reacted with excess of hydrogen, how many liters of water vapor could be produced?
Answers: 3

You know the right answer?
Using the balanced equation N2+O2=2NO, how many grams of NO can be produced when 25.0 grams of N rea...
Questions

English, 07.12.2020 22:30



Chemistry, 07.12.2020 22:30



English, 07.12.2020 22:30

History, 07.12.2020 22:30




Mathematics, 07.12.2020 22:30

English, 07.12.2020 22:30


Mathematics, 07.12.2020 22:30




French, 07.12.2020 22:30