Question 3:
How many atoms are in 25.00 g of B.
A. 1.393 x 10^24 atoms of B
B. 1.333 x...

Chemistry, 10.04.2021 09:10 etxchrissy
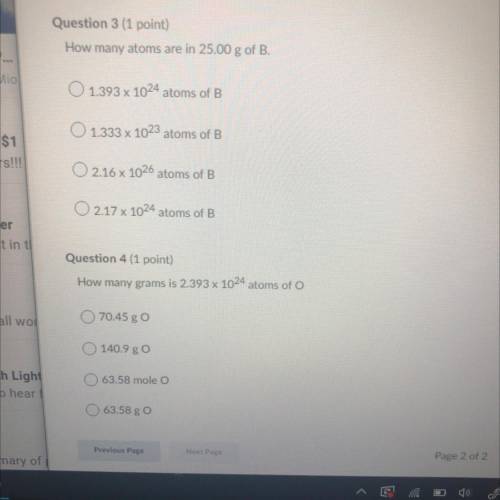
Question 3:
How many atoms are in 25.00 g of B.
A. 1.393 x 10^24 atoms of B
B. 1.333 x 10^23 atoms of B
C. 2.16 x 10^26 atoms of B
D.217 x 10^24 atoms of B
Question 4:
How many grams is 2.393 x 10^24 atoms of O
A. 70.45 g O
B. 140.9 g O
C. 63.58 mole O
D. 63.58 g O
Please answer both questions if you can, thanks for all the efforts :)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 17:10
Some liquids can be distilled, but only at temperatures that are so high that it is impractical, or so high the compound decomposes. explain why distillation such compounds at significantly less than atmospheric pressure (some degree of vacuum) would solve this problem.
Answers: 2

Chemistry, 22.06.2019 19:20
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3
You know the right answer?
Questions


World Languages, 13.10.2021 01:30






Computers and Technology, 13.10.2021 01:30

World Languages, 13.10.2021 01:30

Biology, 13.10.2021 01:30


Biology, 13.10.2021 01:30

Business, 13.10.2021 01:30


Arts, 13.10.2021 01:30

Chemistry, 13.10.2021 01:30

Mathematics, 13.10.2021 01:30



Mathematics, 13.10.2021 01:30



