
Chemistry, 11.04.2021 23:20 kprincess16r
Human blood contains a buffer of carbonic acid (H_2 CO_3) and bicarbonate anion (HC〖O_3〗^-) in order to maintain blood pH between 7.35 and 7.45, as a value higher than 7.8 or lower than 6.8 can lead to death. In this buffer, hydronium and bicarbonate anion are in equilibrium with carbonic acid. Furthermore, the carbonic acid in the first equilibrium can decompose into CO_2 gas and water, resulting in a second equilibrium system between carbonic acid and water. Because CO_2 is an important component of the blood buffer, its regulation in the body, as well as that of O_2 , is extremely important. The effect of this can be important when the human body is subjected to strenuous condition.
Carbonic acid buffer:
H_2 CO_3 (aq)+ H_2 O(l) ↔H_3 O^+ (aq)+HC〖O_3〗^- (aq)
1. Write the equilibrium constant expression K_c for the carbonic acid buffer in the blood.
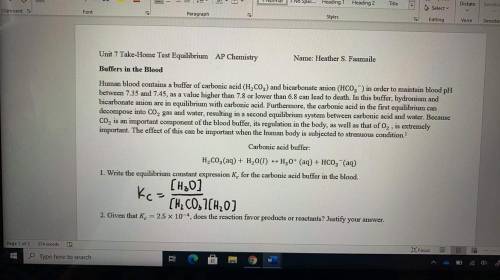

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:30
What is the formula for the molecular compound nitrogen monoxide
Answers: 1

Chemistry, 22.06.2019 03:20
What is the ima of the 1 st class lever in the graphic given? 2 3 0.5
Answers: 1

Chemistry, 22.06.2019 04:30
Long term exposure to waves can cause sunburns and skin cancer. a) visible b) infrared c) gamma rays d) ultraviole
Answers: 1

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3
You know the right answer?
Human blood contains a buffer of carbonic acid (H_2 CO_3) and bicarbonate anion (HC〖O_3〗^-) in order...
Questions



Mathematics, 29.01.2021 17:00


Geography, 29.01.2021 17:00




Physics, 29.01.2021 17:00

Mathematics, 29.01.2021 17:00

Mathematics, 29.01.2021 17:00







Mathematics, 29.01.2021 17:00

Arts, 29.01.2021 17:00

Mathematics, 29.01.2021 17:00



