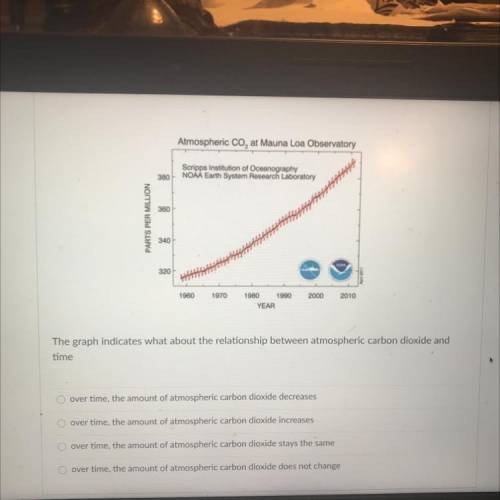
Atmospheric Co, at Mauna Loa Observatory
Scripps Institution of Oceanography
NOAA Earth Syste...

Chemistry, 13.04.2021 04:00 darrenturner
Atmospheric Co, at Mauna Loa Observatory
Scripps Institution of Oceanography
NOAA Earth System Research Laboratory
380
360
PARTS PER MILLION
340
320
1980
1970
2000
2010
1980 1990
YEAR
The graph indicates what about the relationship between atmospheric carbon dioxide and
time
o over time, the amount of atmospheric carbon dioxide decreases
over time, the amount of atmospheric carbon dioxide increases
over time, the amount of atmospheric carbon dioxide stays the same
O over time, the amount of atmospheric carbon dioxide does not change

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:00
Which element in the third period would you expect to have the larger atomic radius, sodium (na) or sulfur (s)? a. sodium, because it has a higher effective nuclear charge attracting electrons in fewer energy levels. b. sodium, because it has fewer protons attracting electrons in the same energy levels. c. sulfur, because it has more protons attracting electrons in more energy levels. d. sulfur, because it has a higher effective nuclear charge attracting electrons in the same energy levels.
Answers: 2

Chemistry, 21.06.2019 21:30
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10^-3 m and k for the dissociation is 1.86x10^-5. ch3cooh(aq)+h2o(> h3o^+(aq)+ch3coo^-(aq)
Answers: 2

Chemistry, 22.06.2019 00:00
Acurium-245 nucleus is hit with a neutron and changes as shown by the equation. complete the equation by filling in the missing parts. 52
Answers: 2

You know the right answer?
Questions



English, 07.07.2020 17:01

Mathematics, 07.07.2020 17:01

Social Studies, 07.07.2020 17:01






History, 07.07.2020 17:01







Social Studies, 07.07.2020 17:01




