
Chemistry, 15.04.2021 14:00 jefersonzoruajas
Hydrochloric acid can be prepared using the reaction described by the chemical equation: 2 NaCl(s) + H2SO4(l) > 2 HCl(g) + Na2 SO4(s). How many grams of HCl can be prepared from 393 g of H2SO4 and 4.00 moles of NaCl?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:30
Un cierto gas tiene un volumen de 800ml a 80°c y 600ml a 80°c y 600mmhg de presión. ¿cual será el volumen del gas a condiciones normales? sí el gas es oxígeno, ¿cuál será su peso? y ¿cuántas moléculas están presentes en el sistema?
Answers: 2

Chemistry, 22.06.2019 03:30
Adrop of acetone (nail polish remover) has a mass of 35 mg and a density of 0.788 g/cm3. what is its volume in cubic centimeters?
Answers: 3

Chemistry, 22.06.2019 05:00
Given sno2 + 2h2 - sn + 2h20 tin oxide reacts with hydrogen to produce tin and water. how many moles of sno2 are needed to produce 500.0 grams of sn?
Answers: 3

Chemistry, 22.06.2019 18:00
The fact that the total amount of energy in a system remains constant is a(n)
Answers: 1
You know the right answer?
Hydrochloric acid can be prepared using the reaction described by the chemical equation: 2 NaCl(s) +...
Questions

English, 27.09.2019 05:30


History, 27.09.2019 05:30

Mathematics, 27.09.2019 05:30



English, 27.09.2019 05:30




Mathematics, 27.09.2019 05:30

History, 27.09.2019 05:30

Mathematics, 27.09.2019 05:30





Health, 27.09.2019 05:30


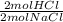 = 4.00 mol HCl
= 4.00 mol HCl


