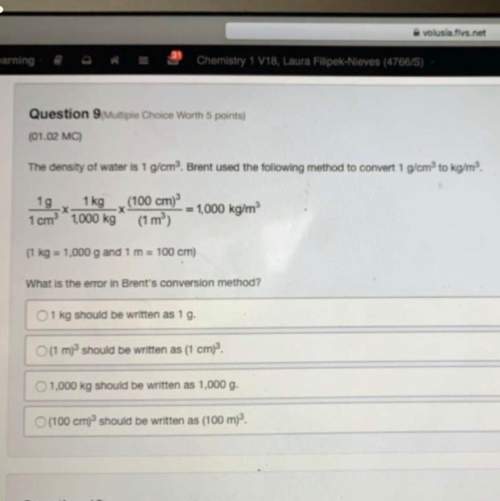
Chemistry, 15.04.2021 22:30 McGregorshamara
2. Consider the carbon atom (ground state configuration 1s22s22p2) and oxygen atom (groundstate configuration 1s22s22p4).(a) Write out the electron configuration (microstate) corresponding to the lowest energyconfiguration of carbon and oxygen. What areLandSin each case?(b) What values ofJare possible with the values ofLandSthat you found for carbon andoxygen in part (a)? Use Hund’s third rule to determine the complete term symbols forthe ground states of carbon and oxygen.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Check the correct box to describe the periodic trends in electronegativity. electronegativity across a period: decreases. increases. electronegativity down a group: decreases. increases.
Answers: 2

Chemistry, 22.06.2019 06:00
In an investigation that uses the scientific method, which step immediately follows making a hypothesis? o summarizing the results o asking a question o making observations designing an experiment mark this and retum save and exit next submit
Answers: 2

Chemistry, 22.06.2019 16:00
Which of the following is the correct definition of chemical energy? a. energy an object has because of its motion or position b. energy resulting from the flow of charged particles, such as electrons or ions c. energy produced from the splitting of atoms d. energy stored in chemical bonds of molecules
Answers: 1

Chemistry, 23.06.2019 02:50
What is the typical rotational frequency frot for a molecule like n2 at room temperature (25∘c)? assume that d for this molecule is 1å=10−10m. take the total mass of an n2 molecule to be mn2=4.65×10−26kg. you will need to account for rotations around two axes (not just one) to find the correct frequency. express frot numerically in hertz, to three significant figures.
Answers: 3
You know the right answer?
2. Consider the carbon atom (ground state configuration 1s22s22p2) and oxygen atom (groundstate conf...
Questions


History, 28.10.2020 14:00

Computers and Technology, 28.10.2020 14:00

Mathematics, 28.10.2020 14:00


Mathematics, 28.10.2020 14:00

Mathematics, 28.10.2020 14:00

Mathematics, 28.10.2020 14:00


History, 28.10.2020 14:00



English, 28.10.2020 14:00

English, 28.10.2020 14:00




Mathematics, 28.10.2020 14:00

Mathematics, 28.10.2020 14:00