
Chemistry, 16.04.2021 01:00 karrathomas
Calcium carbonate decomposes to form calcium oxide and carbon dioxide, like this: (s)(s)(g) At a certain temperature, a chemist finds that a reaction vessel containing a mixture of calcium carbonate, calcium oxide, and carbon dioxide at equilibrium has the following composition: compound amount Calculate the value of the equilibrium constant for this reaction. Round your answer to significant digits.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:40
Salicylic acid is a very important acid. it is used to synthesize the aspirin by treating with acetic anhydride. a 0.2015-g sample of salicylic acid was dissolved in a 100.00-ml volumetric flask, and the solution was diluted to the mark. a 10-ml aliquot of this solution was titrated with standard naoh (0.01130 + 0.2% n) to a phenolphthalein faint pink color end point at 19.81 ml. (a) (calculate the normality of the salicylic acid solution used in the titration. (b) assuming the salicylic acid is pure, what is the equivalent weight of the salicylic acid? practice problems for the final exam (continued) (c) (calculate the inherent error in the determination of the equivalent weight you calculated in part (b). use the following absolute errors in the equipment /glassware when calculating the inherent error. 5.00-ml pipet: + 0.02 ml 100-ml volumetric flask: + 0.08 ml analytical balance: + 0.2 mg 25-ml buret: + 0.03 ml
Answers: 2

Chemistry, 22.06.2019 15:20
Select the most likely product for this reaction: koh(aq) + co2(g) – ? k2co3(aq) + h2o(1) k(s) + h2(g) + o2(g) k(s) + co3(9) +h2
Answers: 2


Chemistry, 23.06.2019 03:30
Calculate the ph of a .10m nh4cl solution. the kb value for nh3 is 1.8×10^-5
Answers: 1
You know the right answer?
Calcium carbonate decomposes to form calcium oxide and carbon dioxide, like this: (s)(s)(g) At a cer...
Questions


Arts, 25.05.2020 07:57

Social Studies, 25.05.2020 07:57



Mathematics, 25.05.2020 07:57

Mathematics, 25.05.2020 07:57





History, 25.05.2020 07:57

Mathematics, 25.05.2020 07:58

Mathematics, 25.05.2020 07:58



Mathematics, 25.05.2020 07:58


Mathematics, 25.05.2020 07:58

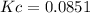
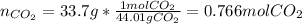
![[CO_2]=\frac{0.766molCO_2}{9.0L}=0.0851M](/tpl/images/1262/6588/3d5ed.png)
![Kc=[CO_2]\\\\Kc=0.0851](/tpl/images/1262/6588/d8c10.png)


