
Chemistry, 16.04.2021 14:00 valeriegarcia12
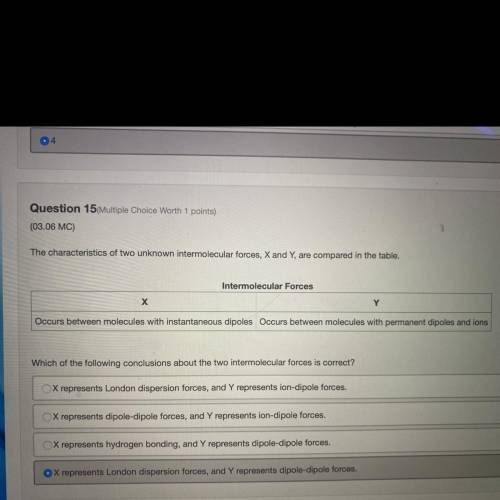
The characteristics of two unknown intermolecular forces, X and Y, are compared in the table.
Intermolecular Forces
х
Y
Occurs between molecules with instantaneous dipoles Occurs between molecules with permanent dipoles and ions
Which of the following conclusions about the two intermolecular forces is correct?
X represents London dispersion forces, and Y represents ion-dipole forces.
X represents dipole-dipole forces, and Y represents ion-dipole forces.
X represents hydrogen bonding, and Y represents dipole-dipole forces.
X represents London dispersion forces, and Y represents dipole-dipole forces.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 08:30
Which common material is an example of a polymer? (25 pts) a. steel b. plastic c. petroleum d. rubbing alcohol
Answers: 2

Chemistry, 22.06.2019 10:00
A50.0g sample of liquid water at 0.0 c ends up as ice at -20.0 c. how much energy is involved in this change?
Answers: 1

Chemistry, 22.06.2019 11:10
Which of the following shapes would represent a molecule with two bonded atoms and 3 lone pairs on only one of them , trigonal planar , bent , trigonal pyramidal , linear
Answers: 1

Chemistry, 22.06.2019 12:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al oxidizing agent = reducing agent =
Answers: 1
You know the right answer?
The characteristics of two unknown intermolecular forces, X and Y, are compared in the table.
Inter...
Questions





Mathematics, 19.05.2020 22:08


English, 19.05.2020 22:08






History, 19.05.2020 22:08

Biology, 19.05.2020 22:08

Mathematics, 19.05.2020 22:08

Health, 19.05.2020 22:08







