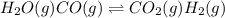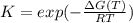Chemistry, 17.04.2021 20:00 andrejr0330jr
For the following exothermic reaction system at equilibrium:
H2O(g) CO(g) CO2(g) H2(g)
Choose the changes that will increase the value of K.
a. Decrease the volume (constant T)
b. Add H2O(g) (constant T)
c. Remove H2(g) (constant T)
d. Add a catalyst (constant T)
e. Add CO2(g) (constant T)
f. Increase the temperature
g. Decrease the temperature

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 09:40
How many grams of aluminum will there be in 98g of al2o3?
Answers: 1

Chemistry, 22.06.2019 12:00
What is the lowest number energy level where a d sublevel is found
Answers: 1

Chemistry, 22.06.2019 14:30
Need ! asap will mark 10 pts using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers
Answers: 1

Chemistry, 22.06.2019 15:00
Which theory was contradicted by experiments with the photoelectric effect? light spreads out after it passes through a small opening. as soon as light strikes metal, electrons will be ejected. visible light, regardless of color, will cause the ejection of electrons when striking metal. the kinetic energy of ejected electrons depends on the frequency of light that strikes the metal.
Answers: 2
You know the right answer?
For the following exothermic reaction system at equilibrium:
H2O(g) CO(g) CO2(g) H2(g)
Choos...
Choos...
Questions

Business, 31.08.2019 04:30

English, 31.08.2019 04:30

History, 31.08.2019 04:30


Mathematics, 31.08.2019 04:30

History, 31.08.2019 04:30


Computers and Technology, 31.08.2019 04:30


History, 31.08.2019 04:30




Arts, 31.08.2019 04:30


English, 31.08.2019 04:30


Social Studies, 31.08.2019 04:30


Mathematics, 31.08.2019 04:30