
Chemistry, 18.04.2021 07:50 alyssakerr17
How many moles of argon gas would be present in a 37.0 liter vessel at 45.00 °C at a pressure of 2.50 atm?
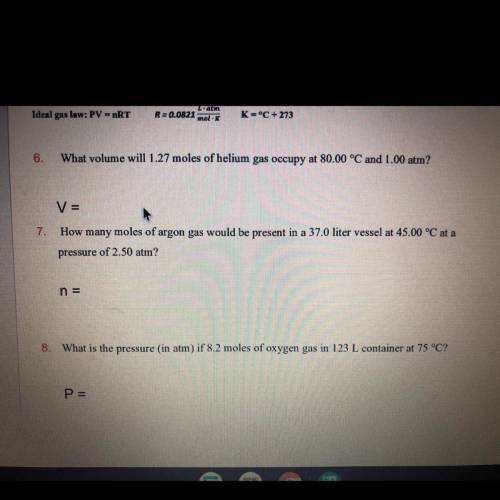

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:10
Using complete sentences, explain how to predict the products and balance the reaction between sulfuric acid and potassium hydroxide.
Answers: 1

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 2

Chemistry, 22.06.2019 19:30
Use the periodic table to find the molar mass of each element. molar mass h = g/mol molar mass s = g/mol molar mass o = g/mol
Answers: 3

Chemistry, 22.06.2019 23:00
What is the measured amount of a product obtained from a chemical reaction?
Answers: 1
You know the right answer?
How many moles of argon gas would be present in a 37.0 liter vessel at 45.00 °C at a pressure of 2.5...
Questions

Mathematics, 20.09.2021 06:50


Mathematics, 20.09.2021 06:50


English, 20.09.2021 06:50



Mathematics, 20.09.2021 06:50


Mathematics, 20.09.2021 06:50






English, 20.09.2021 06:50

Mathematics, 20.09.2021 06:50

Social Studies, 20.09.2021 06:50

Business, 20.09.2021 06:50

History, 20.09.2021 06:50



