Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3


Chemistry, 22.06.2019 12:30
Nebulae are enormous clouds in outer space. they are made mostly of hydrogen gas, helium gas, and dust. some nebulae glow brightly, while others do not. the stars that people see are huge, bright balls of glowing gas. they are made mostly of hydrogen and helium. which statement correctly describes other ways in which nebulae and stars are different? a. stars can form inside a nebula but a nebula can never be produced by any star. b. a star always has a higher density than a nebula. c. stars can never form inside a nebula but a nebula can be produced by any star. d. a nebula always has a higher density than a star.
Answers: 3

Chemistry, 22.06.2019 14:30
Need ! asap will mark 10 pts using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers
Answers: 1
You know the right answer?
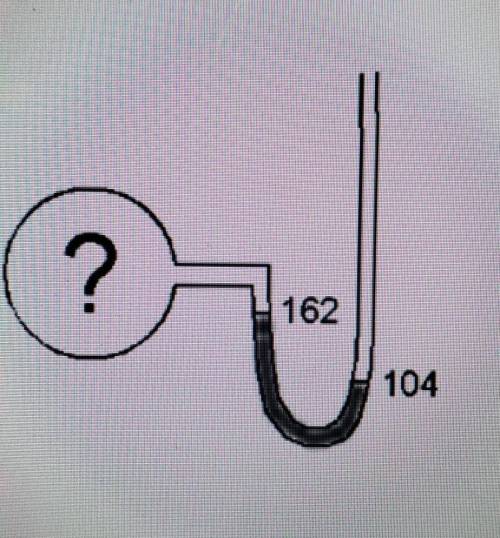
The diagram at right shows a manometer connected to a flask. The mercury levels are measured in mm....
Questions



History, 25.11.2021 15:40


Advanced Placement (AP), 25.11.2021 15:40

World Languages, 25.11.2021 15:40

Mathematics, 25.11.2021 15:40

English, 25.11.2021 15:40



Physics, 25.11.2021 15:40

Biology, 25.11.2021 15:40


Chemistry, 25.11.2021 15:40

Biology, 25.11.2021 15:40

Biology, 25.11.2021 15:40

Business, 25.11.2021 15:40


Mathematics, 25.11.2021 15:40

Business, 25.11.2021 15:40