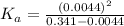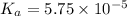Chemistry, 19.04.2021 16:10 NarutoBeast8049
In the laboratory, a general chemistry student measured the pH of a 0.341 M aqueous solution of benzoic acid, C6H5COOH to be 2.351. Use the information she obtained to determine the Ka for this acid.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:30
The table lists pressure and volume values for a particular gas. which is the best estimate for the value of v at p = 7.0 × 103 pascals?
Answers: 3

Chemistry, 22.06.2019 12:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al oxidizing agent = reducing agent =
Answers: 1

Chemistry, 22.06.2019 14:00
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3

Chemistry, 23.06.2019 03:30
Scientists often deal with numbers that are either very large or very small. for example, the radius of the sun is approximately 696,000 kilometers, while bacterial cells are as small as 1.9 × 10-4 millimeters. express each of these numbers in an alternate form.
Answers: 3
You know the right answer?
In the laboratory, a general chemistry student measured the pH of a 0.341 M aqueous solution of benz...
Questions

Mathematics, 06.12.2021 05:20


Mathematics, 06.12.2021 05:30

History, 06.12.2021 05:30

Chemistry, 06.12.2021 05:30






Chemistry, 06.12.2021 05:30



Law, 06.12.2021 05:30


English, 06.12.2021 05:30

Mathematics, 06.12.2021 05:30

Mathematics, 06.12.2021 05:30


History, 06.12.2021 05:30

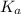 for the acid is
for the acid is 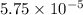
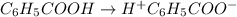
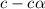
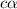
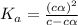
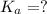
![pH=-log[H^+]](/tpl/images/1268/1073/15713.png)
![[H^+]=10^{-2.351}=0.0044](/tpl/images/1268/1073/85ea6.png)
![[H^]=c\alpha=0.0044](/tpl/images/1268/1073/4f06a.png)