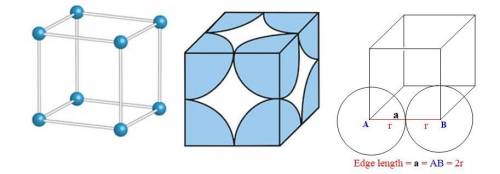
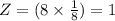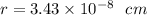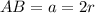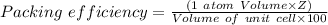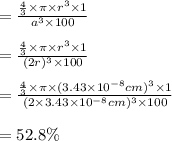The different unit cell types have a different packing efficiency. The simple cubic has the least efficient packing and the face-centered cubic has the most efficient packing (sometimes the face-centered cubic is called cubic closest packing). You can determine what percent of the unit cell is occupied by (1) determining the volume of the whole unit cell and (2) determining the volume of the occupied space by the atoms in the unit cell. Remember that the volume of a cube is V

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:00
From the options provided for each element below, choose the properties that it may have based on its location in the periodic table fluorine (f): highly reactive nonmetal shiny a conductor
Answers: 1


Chemistry, 22.06.2019 20:00
Listenbase your answer to the question on the information below.nuclear radiation is harmful to living cells, particularly to fast-growing cells, such as cancer cells and blood cells. an external beam of the radiation emitted from a radioisotope can be directed on a small area of a person to destroy cancer cells within the body.cobalt-60 is an artificially produced radioisotope that emits gamma rays and beta particles. one hospital keeps a 100.0-gram sample of cobalt-60 in an appropriate, secure storage container for future cancer treatment.which choice represents the correct product for the beta decay of the co-60? fe-60ni-60fe-61ni-61
Answers: 2

Chemistry, 22.06.2019 22:00
Pls ill give u brainliest which of the following is true about science? 1. political conditions are unable to influence it. 2. economic concerns may prevent it from solving problems.
Answers: 2
You know the right answer?
The different unit cell types have a different packing efficiency. The simple cubic has the least ef...
Questions


Biology, 03.08.2019 16:00


History, 03.08.2019 16:00

Social Studies, 03.08.2019 16:00

History, 03.08.2019 16:00


Mathematics, 03.08.2019 16:00




Mathematics, 03.08.2019 16:00



English, 03.08.2019 16:00

History, 03.08.2019 16:00

Mathematics, 03.08.2019 16:00

Mathematics, 03.08.2019 16:00


English, 03.08.2019 16:00

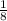 to both the cell unit
to both the cell unit