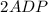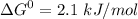Write balanced equations for all the reactions in the catabolism of glucose to two molecules of glyceraldehyde 3-phosphate (the preparatory phase of glycolysis), including the standard free-energy change for each reaction. Then write the overall or net equation for the preparatory phase of glycolysis, with the net standard free-energy change.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3

Chemistry, 22.06.2019 10:10
Stage in which a typical star has completely stopped fusion
Answers: 1

Chemistry, 22.06.2019 20:30
Which of the following is not true about the atomic model of substances?
Answers: 1

You know the right answer?
Write balanced equations for all the reactions in the catabolism of glucose to two molecules of glyc...
Questions


Mathematics, 21.05.2021 14:00



History, 21.05.2021 14:00


English, 21.05.2021 14:00

English, 21.05.2021 14:00


English, 21.05.2021 14:00


Business, 21.05.2021 14:00

Arts, 21.05.2021 14:00



Mathematics, 21.05.2021 14:00


Mathematics, 21.05.2021 14:00

Social Studies, 21.05.2021 14:00

Chemistry, 21.05.2021 14:00

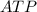 → glucose -
→ glucose - 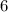 - phosphate
- phosphate 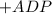 ,
, 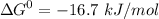
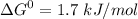
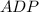 + Fructose -
+ Fructose - 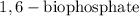 ,
, 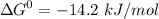
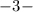 phosphate,
phosphate, 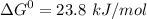
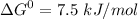
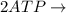 glyceraldehyde
glyceraldehyde