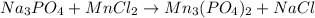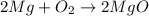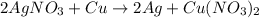Chemistry, 11.10.2019 07:00 Sydney012618
Which of the following demonstrates a double-replacement reaction?
a: na3po4 + mncl2 > mn3(po4)2 + nacl
b: caco3 > cao + co2
c: 2mg + o2 > 2mgo
d: 2agno3 + cu > 2ag + cu(no3)2

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 13:30
If the concentration of phosphate in the cytosol is 2.0 mm and the concentration of phosphate in the surrounding fluid is 0.1 mm, how could the cell increase the concentration of phosphate in the cytosol? a) passive transportb) diffusionc) active transportd) osmosise) facilitated diffusion
Answers: 3

Chemistry, 23.06.2019 06:10
How much would the freezing point of water decrease if 4 mol of nacl were added to 1 kg of water (kf=1.86 degrees c/(mol/kg) for water and i=2 for nacl a- 7.44 degrees c b- 14.88 c 3.72 d 1.86
Answers: 1

Chemistry, 23.06.2019 08:00
Match the vocabulary terms to their definitions. 1 . a long, chain-like set of molecules made up of repeating units joined end to end polymer 2 . a hard, brittle, heat- and corrosion-resistant material made by subjecting a nonmetallic mineral mixture to intense heat ceramic 3 . a plastic with low elongations that cannot be recycled thermoset 4 . a carbon fiber embedded in a polymer resin matrix thermoplastic 5 . a plastic with high elongations that can be recycled crystal 6 . a solid form resulting from the arrangement of atoms, ions, or molecules in definite geometric patterns composite
Answers: 1

Chemistry, 23.06.2019 11:00
Nh4no3 n2o + 2h2o a chemist who is performing this reaction starts with 160.1 g of nh4no3. the molar mass of nh4no3 is 80.03 g/mol; the molar mass of water (h2o) is 18.01 g/mol. what mass, in grams, of h2o is produced?
Answers: 1
You know the right answer?
Which of the following demonstrates a double-replacement reaction?
a: na3po4 + mncl2 > m...
a: na3po4 + mncl2 > m...
Questions

Mathematics, 22.08.2019 05:50

Mathematics, 22.08.2019 05:50




Mathematics, 22.08.2019 05:50

Social Studies, 22.08.2019 05:50




Mathematics, 22.08.2019 05:50

Mathematics, 22.08.2019 05:50

History, 22.08.2019 05:50

Social Studies, 22.08.2019 05:50





Mathematics, 22.08.2019 05:50

History, 22.08.2019 05:50