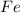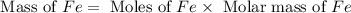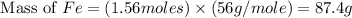Chemistry, 24.11.2019 12:31 briannagisellegarcia
Fe2o3 + 2al -> al2o3 + 2fe
calculate the mass of iron metal (in grams) that can be prepared from 150 grams of aluminum and 250 grams of iron(iii) oxide.
!

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:30
Each pair of clay balls represents to planetesimals if each plane test molluscum pound of the same material and is separated by the same distance which pair experiences the greatest gravitational attraction
Answers: 2

Chemistry, 22.06.2019 08:00
Identify a strong intermolecular force of attraction between an alcohol
Answers: 1

Chemistry, 22.06.2019 16:00
Sulfuric acid is a polyprotic acid. write balanced chemical equations for the sequence of reactions that sulfuric acid can undergo when it's dissolved in water.
Answers: 2

Chemistry, 23.06.2019 03:00
Air pressure is measured in pascals. for a professional american football game, the ball should be inflated to about 90,000 pascals. scientists studied the effects of air temperature on the pressure inside american footballs by taking these steps: 1. prepare 100 footballs. 2. measure each football's air pressure. 3. divide footballs into 10 groups. 4. place the groups in different lockers cooled to different air temperatures. 5. after 12 hours, remove the footballs from lockers. 6. measure each football's pressure again. 7. compare the new pressures to the starting pressures. what two terms best describe the variable "air pressure inside the football" in this experiment? independent, qualitative independent, quantitative dependent, qualitative dependent, quantitative
Answers: 3
You know the right answer?
Fe2o3 + 2al -> al2o3 + 2fe
calculate the mass of iron metal (in grams) that can be p...
calculate the mass of iron metal (in grams) that can be p...
Questions

History, 26.08.2019 06:30

Mathematics, 26.08.2019 06:30

Chemistry, 26.08.2019 06:30



Mathematics, 26.08.2019 06:30

Biology, 26.08.2019 06:30

History, 26.08.2019 06:30

Computers and Technology, 26.08.2019 06:30

Chemistry, 26.08.2019 06:30




Computers and Technology, 26.08.2019 06:30


History, 26.08.2019 06:30


Physics, 26.08.2019 06:30


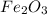 = 250 g
= 250 g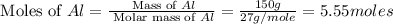
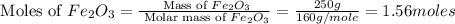
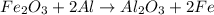
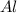 react with 1 mole of
react with 1 mole of 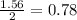 moles of
moles of