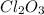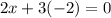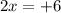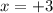Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:20
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1

Chemistry, 22.06.2019 04:50
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 13:20
Can someone me with 3 and 4 plz. this is for masteries test.
Answers: 2

Chemistry, 22.06.2019 18:00
To apply in a gold the individual gold atoms are united to each other by means of a metallic bond. how would you use the gold block to determine the atomic radius of a gold atom?
Answers: 3
You know the right answer?
What is the oxidation number of cl in cl2o3...
Questions



Chemistry, 29.08.2019 11:10

Mathematics, 29.08.2019 11:10


Biology, 29.08.2019 11:10

Mathematics, 29.08.2019 11:10




Mathematics, 29.08.2019 11:10


Biology, 29.08.2019 11:10

Social Studies, 29.08.2019 11:10



Arts, 29.08.2019 11:10

Mathematics, 29.08.2019 11:10

Mathematics, 29.08.2019 11:10

Mathematics, 29.08.2019 11:10