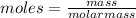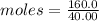Chemistry, 05.02.2020 13:50 uglybruja81
Asample of sodium hydroxide (naoh) has a mass of 160.0 g. the molar mass of naoh is 40.00 g/mol. how many moles of naoh does this sample contain?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:00
Write a hypothesis that answers the lesson question, “while observing a chemical reaction, how can you tell which reactant is limiting? ” hypothesis: if a substance is the limiting reactant, then . . because . .
Answers: 1

Chemistry, 22.06.2019 12:00
What does a complete balanced chemical equation include? a. exothermic coefficients b. endothermic coefficients c. valence electrons d. molar coefficients
Answers: 1

Chemistry, 22.06.2019 14:30
Is a pencil falling to the floor anon contact force, a force, or a contact force
Answers: 1

Chemistry, 22.06.2019 23:00
Movement that is like a t a type of wave that transfers energy where the particles in the medium move in a circle motion while the energy travels left or right. a type of wave that transfers energy where the particles in the medium move perpendicular to the direction in which the energy is traveling. transfers energy from one location to another a type of wave that transfers energy where the particles in the medium move parallel to the direction in which the energy is traveling. movement that is back and forth, like an equal sign = 1. wave 2. parallel movement 3. perpendicular movement 4. transverse wave 5. longitudinal wave 6. surface wave
Answers: 1
You know the right answer?
Asample of sodium hydroxide (naoh) has a mass of 160.0 g. the molar mass of naoh is 40.00 g/mol. how...
Questions

Chemistry, 02.11.2020 18:30




Mathematics, 02.11.2020 18:30



Mathematics, 02.11.2020 18:30

English, 02.11.2020 18:30


Biology, 02.11.2020 18:30



Chemistry, 02.11.2020 18:30





History, 02.11.2020 18:30