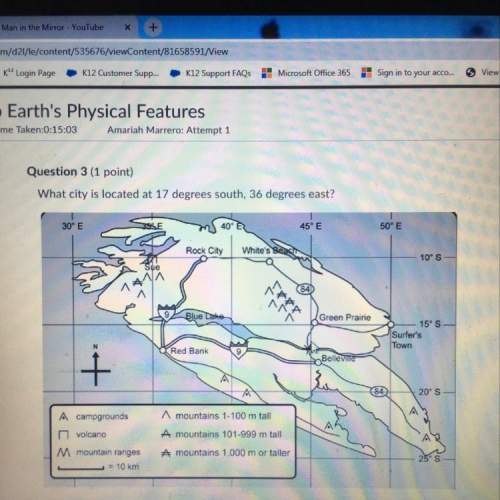
Which applies to the collision theory?
a. particles need to collide in order to react.<...

Chemistry, 30.01.2020 12:50 tristantisdale1
Which applies to the collision theory?
a. particles need to collide in order to react.
b. particles may obtain successful collisions in any molecular orientation.
c. temperature increase causes kinetic energy of particles to decrease.
d. reactions do not have to begin with collision of molecules or particles.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 14:30
Which of the following represents the ester functional group? a. -coo- b. -cho c. -cooh d. c=o
Answers: 1

Chemistry, 22.06.2019 20:50
What is the vapor pressure of a solution with a benzene to octane?
Answers: 2

Chemistry, 22.06.2019 23:00
Which of your 24 wells had indications that a chemical reaction occurred? how were you able to tell that a chemical reaction occurred? which of your 24 wells had indications that a physical reaction occurred? how were you able to tell that a physical reaction occurred? report on both mixing and evaporation. make a general statement about whether your hypotheses were validated or rejected. must your hypotheses be correct for this to be a successful laboratory?
Answers: 3

You know the right answer?
Questions



Mathematics, 05.02.2022 14:00

Mathematics, 05.02.2022 14:00

English, 05.02.2022 14:00

Biology, 05.02.2022 14:00

Mathematics, 05.02.2022 14:00

History, 05.02.2022 14:00






English, 05.02.2022 14:00

Biology, 05.02.2022 14:00

English, 05.02.2022 14:00




English, 05.02.2022 14:00