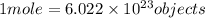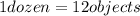How are a mole and a dozen similar?
a. each represents a certain mass of material.
b....

Chemistry, 26.08.2019 00:20 stephesquilin
How are a mole and a dozen similar?
a. each represents a certain mass of material.
b. each names a given number of objects.
c. each identifies a specific volume.
d. each measures a given number of amus.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Reaction rate depends on how many molecules are coming into contact with each other with enough energy to react. increasing the temperature of the reactants will increase -
Answers: 3

Chemistry, 22.06.2019 03:50
Which of the following statements about acidic water is true? a. acid has no effect on the h,o molecules. b. the solution contains a larger number of oh ions than h,o ions. c. the solution contains a larger number of h,o ions than qh ions. d. the solution contains an equal number of h,o ions and oh ions. none of the above e.
Answers: 1

Chemistry, 22.06.2019 06:00
One of the few xenon compounds that form is cesium xenon heptafluoride (csxef7). how many moles of csxef7 can be produced from the reaction of 13.0 mol cesium fluoride with 12.5 mol xenon hexafluoride? csf(s) + xef6(s) csxef7(s)
Answers: 1

Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3
You know the right answer?
Questions


Social Studies, 01.04.2021 21:10


Mathematics, 01.04.2021 21:10



English, 01.04.2021 21:10


Advanced Placement (AP), 01.04.2021 21:10


Arts, 01.04.2021 21:10

Mathematics, 01.04.2021 21:10

Mathematics, 01.04.2021 21:10


Social Studies, 01.04.2021 21:10





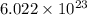 .
.