Could someone confirm that this is correct? !
...

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1


Chemistry, 22.06.2019 19:00
Imagine that a new planet is discovered with two moons of equal mass: moon a and moon b. the mass of the new planet is greater than the combined mass of its moons. moon a is farther away from the new planet than moon b. what is the planet's gravitational pull on moon a compared to the planet's gravitational pull on moon b? the planet's gravity repels moon a with a greater force than it repels moon b, which is why moon a is farther away. the gravitational pull on moon b is greater than on moon a because moon b is closer to the new planet than moon a. the gravitational pull on moon b is greater than on moon a because moon b is farther away from the new planet than moon a. the gravitational pull on moon a is the same as the gravitational pull on moon b because distance does not affect the planet's gravity.
Answers: 1

Chemistry, 22.06.2019 22:30
Calculate the concentration of all species in a 0.165 m solution of h2co3.
Answers: 1
You know the right answer?
Questions

Biology, 01.09.2020 21:01

History, 01.09.2020 21:01

Mathematics, 01.09.2020 21:01

History, 01.09.2020 21:01

Mathematics, 01.09.2020 21:01

Chemistry, 01.09.2020 21:01

Mathematics, 01.09.2020 21:01

English, 01.09.2020 21:01

English, 01.09.2020 21:01

English, 01.09.2020 21:01

Mathematics, 01.09.2020 21:01


Mathematics, 01.09.2020 21:01


Mathematics, 01.09.2020 21:01


Biology, 01.09.2020 21:01


Social Studies, 01.09.2020 21:01

English, 01.09.2020 21:01


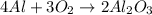
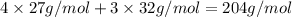
![2\times 102g/mol=204g/mol]](/tpl/images/0274/8461/0666f.png)



