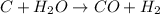Chemistry, 12.01.2020 19:31 ausemkattom3034
How many grams of h2 would be formed if 34 grams of carbon reacted with an unlimited amount of h2o? the reaction is:
c + h2o → co + h2
the atomic mass of c is 12.01 g/mole. the atomic mass of h2 is 2.016 g/mole. finish the problem by choosing the correct format for dimensional analysis.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:30
In a reaction (at equilibrium) that makes more moles of gas than it consumes, what is the effect of increasing the pressure?
Answers: 1

Chemistry, 22.06.2019 10:00
Ahydrogen atom has 1 electron. how many bonds can hydrogen form? a) 1 b) 2 c) 3 d) 4 e) 5
Answers: 3

Chemistry, 22.06.2019 16:00
Which factor is likely to impact the possible number of compounds ?
Answers: 1

Chemistry, 22.06.2019 20:30
Select all the correct answers.which compounds have the empirical formula ch20? (multiple answers)a.c2h4o2b.c3h603c.ch2o2d.c5h1005e.c6h1206
Answers: 2
You know the right answer?
How many grams of h2 would be formed if 34 grams of carbon reacted with an unlimited amount of h2o?...
Questions




















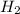 will be, 5.66 grams
will be, 5.66 grams