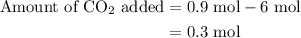Chemistry, 30.01.2020 05:57 Queiao4088
An equilibrium mixture contains 0.600 mol of each of the products (carbon dioxide and hydrogen gas) and 0.200 mol of each of the reactants (carbon monoxide and water vapor) in a 1.00-l container. how many moles of carbon dioxide would have to be added at constant temperature and volume to increase the amount of carbon monoxide to 0.300 mol once equilibrium has been reestablished?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 09:30
Mike and mitchell decide to have a foot race. they mark off a stretch of 100 yards, and recruit cindy to work the stopwatch. after running the race and looking at the results, cindy declared that mitchell was the fastest. so how did the boys times compare?
Answers: 3

Chemistry, 22.06.2019 11:30
Compare and contrast refraction of light and sound will give brainliest
Answers: 1

Chemistry, 22.06.2019 14:00
Which of the following is true about a carbonated soft drink? . the carbon dioxide is the solvent, and water is the solute.. the water is the solution, and carbon dioxide is the solvent.. the carbon dioxide is the solution, and the water is the solvent.. the water is the solvent, and the carbon dioxide is the solute.. .
Answers: 1

Chemistry, 22.06.2019 22:00
What mass of glucose is produced when 54g of water react with carbon dioxide
Answers: 1
You know the right answer?
An equilibrium mixture contains 0.600 mol of each of the products (carbon dioxide and hydrogen gas)...
Questions


Mathematics, 28.08.2020 01:01

Mathematics, 28.08.2020 01:01


Chemistry, 28.08.2020 01:01

Mathematics, 28.08.2020 01:01

Chemistry, 28.08.2020 01:01











Mathematics, 28.08.2020 01:01

Social Studies, 28.08.2020 01:01

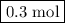 of
of 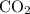 are added so as to increase the amount of carbon monoxide to 0.3 mol.
are added so as to increase the amount of carbon monoxide to 0.3 mol.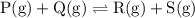
![{\text{K}}=\dfrac{{\left[ {\text{R}} \right]\left[ {\text{S}}\right]}}{{\left[{\text{P}}\right]\left[ {\text{Q}} \right]}}](/tpl/images/0484/7673/9b899.png)
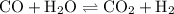
![{\text{K = }}\dfrac{{\left[ {{\text{C}}{{\text{O}}_{\text{2}}}} \right]\left[{{{\text{H}}_{\text{2}}}} \right]}}{{\left[ {{\text{CO}}}\right]\left[{{{\text{H}}_2}{\text{O}}} \right]}}](/tpl/images/0484/7673/6dcad.png) .......(1)
.......(1)![\left[{{\text{C}}{{\text{O}}_{\text{2}}}}\right]](/tpl/images/0484/7673/9014c.png) is the concentration of carbon dioxide.
is the concentration of carbon dioxide.
![\left[{{{\text{H}}_{\text{2}}}}\right]](/tpl/images/0484/7673/340fe.png) is the concentration of hydrogen.
is the concentration of hydrogen.
![\left[{{\text{CO}}}\right]](/tpl/images/0484/7673/d6da7.png) is the concentration of carbon monoxide.
is the concentration of carbon monoxide.
![\left[{{{\text{H}}_2}{\text{O}}}\right]](/tpl/images/0484/7673/62a9e.png) is the concentration of water.
is the concentration of water.
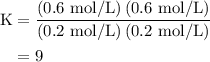
![\left[{{\text{C}}{{\text{O}}_{\text{2}}}}\right]=\dfrac{{{\text{K}}\left( {\left[{{\text{CO}}} \right]\left[{{{\text{H}}_2}{\text{O}}}\right]}\right)}}{{\left[{{{\text{H}}_{\text{2}}}} \right]}}](/tpl/images/0484/7673/f6940.png) ......(2)
......(2)![\begin{aligned}\left[ {{\text{C}}{{\text{O}}_{\text{2}}}}\right]&= \frac{{{\text{9}}\left( {{\text{0}}{\text{.3 mol/L}}}\right)\left({{\text{0}}{\text{.2 mol/L}}}\right)}}{{{\text{0}}{\text{.6 mol/L}}}}\\&= 0.{\text{9 mol/L}}\\\end{aligned}](/tpl/images/0484/7673/95ec6.png)