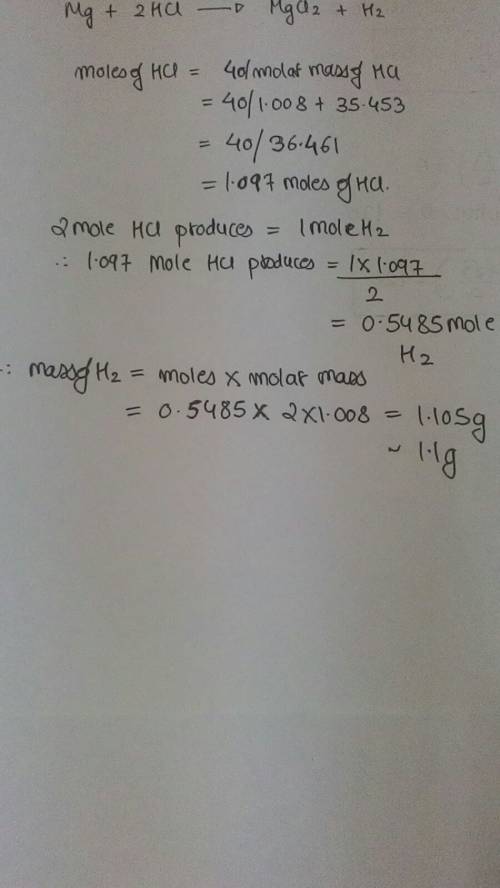Chemistry, 16.11.2019 04:31 idontknow113
If 40.0 g of hcl react with an excess of magnesium metal, what is the theoretical yield of hydrogen?
1.11 g
2.22 g
52.2 g
104 g

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 19:00
Mercury metal is poured into a graduated cylinder that holds exactly 22.5 ml the mercury used to fill the cylinder mass in 306.0 g from this information calculate the density of mercury
Answers: 2

Chemistry, 23.06.2019 15:00
For part 1, describe the changes in the colors of the well, if any, as you go from well 1 to well 9—that is, as you go from the well with the least copper(ii) nitrate to the well with the most copper(ii) nitrate. which wells had the most distinct precipitate? for part 2, describe the changes in the colors of the well, if any, as you go from well 1 to well 9—that is, as you go from the well with the least iron(ii) sulfate to the well with the most iron(ii) sulfate. which wells had the most distinct precipitate? for part 3, describe the changes in the colors of the well, if any, as you go from well 1 to well 9—that is, as you go from the well with the least iron(iii) nitrate to the well with the most iron(iii) nitrate. which wells had the most distinct precipitate?
Answers: 3

Chemistry, 23.06.2019 15:40
The poh of a solution is 6.0. which statement is correct? use poh=-log[oh and ph+poh= 14 o o o the ph of the solution is 20.0. the concentration of oh ions is 1.0 x 10-8 m- the concentration of oh ions is 1.0 x 106 m- the ph of the solution is 8.0.
Answers: 3
You know the right answer?
If 40.0 g of hcl react with an excess of magnesium metal, what is the theoretical yield of hydrogen?...
Questions

Mathematics, 26.04.2021 22:50



Mathematics, 26.04.2021 22:50





Mathematics, 26.04.2021 22:50

Mathematics, 26.04.2021 22:50

Mathematics, 26.04.2021 22:50

History, 26.04.2021 22:50


Mathematics, 26.04.2021 22:50

Mathematics, 26.04.2021 22:50

Mathematics, 26.04.2021 22:50

English, 26.04.2021 22:50

Computers and Technology, 26.04.2021 22:50