
Chemistry, 09.01.2020 08:31 keigleyhannah30
In the first law of thermodynamics, △e = q-w, what does △e stand for?
a. the work done by the piston
b. the internal energy of the system
c. the heat added to the system
d. the change in the internal energy of the system

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 18:00
What amount of heat is exchanged when 106.2 grams of substance y goes from a liquid at 35 degrees celsius to a solid at the same temperature? melting point of substance y = 35 degrees c; δhvaporization = 3.67 j/mol; δhfusion = 3.30 j/mol. mwsubstance y = 28.22 g/mol. −12.4 j −3.51 x 102 j 1.24 x 101 j 351 j
Answers: 1

Chemistry, 23.06.2019 03:30
Mr. rose asked his student to draw a quadrilateral with four unequal sides. an example of this kind of quadrilateral
Answers: 1

Chemistry, 23.06.2019 05:00
C=59(f−32)the equation above shows how temperature f, measured in degrees fahrenheit, relates to a temperature c, measured in degrees celsius. based on the equation, which of the following must be true? a temperature increase of 1 degree fahrenheit is equivalent to a temperature increase of 59 degree celsius.a temperature increase of 1 degree celsius is equivalent to a temperature increase of 1.8 degrees fahrenheit.a temperature increase of 59 degree fahrenheit is equivalent to a temperature increase of 1 degree celsius.a) i onlyb) ii onlyc) iii onlyd) i and ii only
Answers: 1
You know the right answer?
In the first law of thermodynamics, △e = q-w, what does △e stand for?
a. the work done...
a. the work done...
Questions

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Social Studies, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

Mathematics, 11.09.2020 14:01

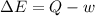
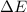 represents the internal energy of the system.
represents the internal energy of the system.

