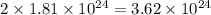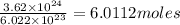Chemistry, 04.02.2020 18:01 shongmadi77
Calculate the number of molecules in 7.00 moles h2s.
calculate the number of moles of cl atoms in 1.81 x10^24 units of magnesium chloride, mgcl2.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 23.06.2019 04:10
An unknown substance has been shown to have metallic bonds. which of the following is most likely a property of this substance? a. low conductivity b. low boiling point c. high malleability d. high solubility in water
Answers: 2

Chemistry, 23.06.2019 11:30
Which of these have the same number of particles as 1 mole of water h2o
Answers: 1
You know the right answer?
Calculate the number of molecules in 7.00 moles h2s.
calculate the number of moles of c...
calculate the number of moles of c...
Questions


History, 04.10.2019 22:00



Health, 04.10.2019 22:00



Chemistry, 04.10.2019 22:00




Physics, 04.10.2019 22:00



Social Studies, 04.10.2019 22:00


Health, 04.10.2019 22:00

Spanish, 04.10.2019 22:00

Mathematics, 04.10.2019 22:00

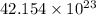
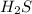 contains
contains 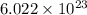 number of molecules
number of molecules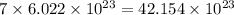 number of molecules
number of molecules